Bioelectrochemical systems (BESs) are electrochemical cells that use microorganisms as the catalyst on one or both electrodes. The reactions occurring at the electrodes are catalyzed by microorganisms, while at the same time electrons are transferred from the oxidized substrate to the anode, or electrons are accepted from the cathode for a reduction reaction. BES has been deemed as an attractive treatment method due to continuous nutrient enrichment and energy recovery as electricity (microbial fuel cell, MFC, Fig.1) or value-added products such as hydrogen gas (microbial electrolysis cell, MEC, Fig. 2). Alternatively, BES system can be applied in desalination, known as microbial desalination cell (MDC), by inserting a third chamber made from ion exchange membrane between anode and cathode (Fig. 3).
Figure 1. Glucose serves as an example fuel. a | An indirect microbial fuel cell. A fermentative microorganism converts glucose to an end product, hydrogen, which can abiotically react with the anode to produce electrons and protons. This process only partially recovers the electrons available in the organic fuel as electricity, and results in the accumulation of organic products in the anode chamber. b | A mediator-driven microbial fuel cell. An electron-shuttling mediator accepts electrons from reduced cell constituents and abiotically transfers the electrons to the anode. The reoxidized mediator can then undergo repeated cycles of reduction and oxidation. In most instances, the cells that have been used in such fuel cells only incompletely oxidize their organic fuels as shown. c | The oxidation of glucose to carbon dioxide with direct electron transfer to the electrode surface. Glucose is taken into the cell and oxidized to carbon dioxide by typical central metabolic pathways, such as the tricarboxylic acid (TCA) cycle. Electrons derived from glucose oxidation are transferred across the inner membrane, periplasm, and outer membrane through electron transport proteins, such as c-type cytochromes. In this example, the system is illustrated with an air cathode rather than a cathode submerged in water. d | A two-chambered microbial fuel cell. This system is not optimized for maximum power production but is convenient for microbiological studies. Click on the figure to find out the original source.
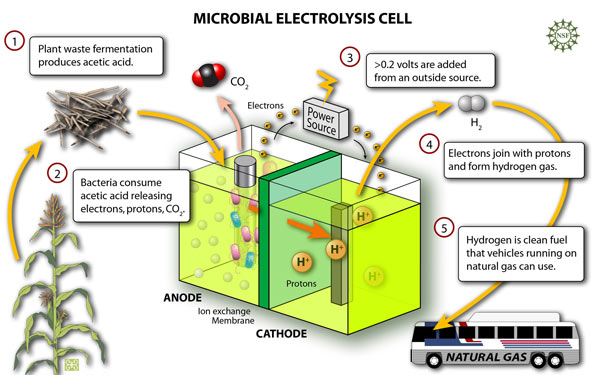
Figure 2. The schematic and potential application of microbial electrolysis cell. Click on the figure to find out the original source.
Figure 3. A general schematic of MDC for saline water desalination purpose. Click on the figure to find out the original source.